Research We’re Reading: From the desks of Martin Kohlmeier, MD, PhD and Robyn Amos-Kroohs, PhD
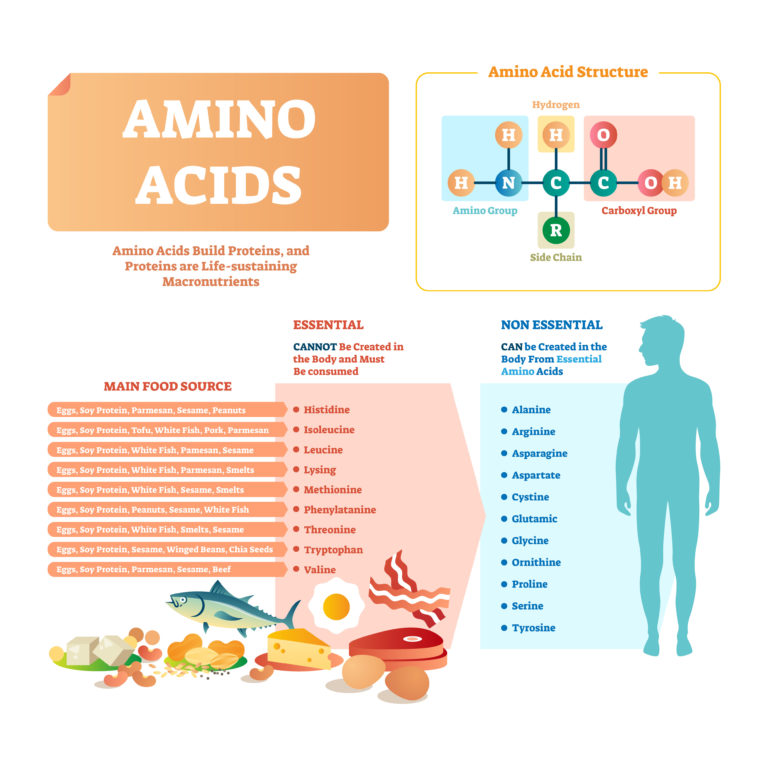
August 28, 2017 – Protein is an important part of every cell in the body. Protein is also a building block of enzymes, hormones, and other important substances used in body processes. It’s a major component of most body systems, including the immune system, metabolism, and circulatory system. Its importance is why protein is known as a macronutrient, meaning that large amounts are required to help the body function appropriately on a daily basis. And unlike sugar and fats, macronutrients that have acquired bad reputations, protein is recognized as an important part of a healthy diet.
Most of us gather the majority of this vital nutrient from animal sources, such as eggs, meat, and dairy products. Animal protein has the advantage of being a complete protein source, meaning that it supplies all of the 22 amino acids that are required in the body on a daily basis, including nine essential amino acids that the body can’t make on its own. The importance of adequate daily intake cannot be overstated, because your body doesn’t store protein long term the way it does fats and carbohydrates.
For health or personal reasons, however, many people choose to exclude animal protein from their diets. Instead, they source their proteins from vegetables, legumes, and nuts or seeds. Plant proteins don’t always have the full amount of all essential amino acids, but combinations, such as beans and corn, tend to balance each other. Therefore, the key for a healthy plant-based diet is variety and knowing about good plant protein combinations.
Whether sourced from animals or plants, the body uses protein to build new muscle and repair old tissue. There has been some question, however, about whether the source of the protein affects muscle strength and musculoskeletal system health. More and stronger muscles are not just what body builders and athletes want, they also ensure more robust health, important with advancing age. Since everyone can benefit from increased muscle and bone health regardless of their diet preferences, researchers have recently assessed whether animal- or plant-sourced protein makes a difference in muscle strength and health.
In this study, researchers used a validated food frequency questionnaire (FFQ) to assess typical dietary intakes of foods and nutrients. In particular, they calculated how much protein from all food sources participants ate, then analyzed the findings by different types of protein. Participants also completed questionnaires about their levels of physical activity, from being asleep to being very active. The two questionnaires about food and activity, along with body composition and muscle strength, were analyzed to determine relationships between the data. Examination of food patterns and muscle health in these middle-aged men and women found that higher protein intake went with greater muscle mass and strength. The differences were small overall (2-3%) and most of that between the lowest and the next protein intake quartile (medians 0.8 g/kg vs 1.1 g/kg). When participants were clustered by the types of protein sources in their diet, muscle measures were similar. It did not seem to matter whether protein came mainly from plant sources, or from meat, dairy products, or fish. These findings appear to support the notion that adults can sustain healthy and strong muscles with a plant-based diet just as well as with meats, fish, or dairy foods. It’s not important how you get your protein from your diet, but it is important that you get enough of it.
Source: Mangano KM et al. Dietary protein is associated with musculoskeletal health independent of dietary pattern. The Framingham Third Generation Family Study. Am J Clin Nutr 2017 105(3):714-722